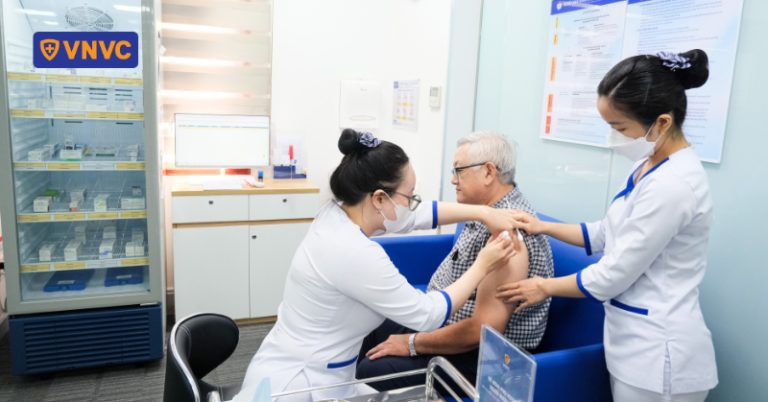
On December 14, a major vaccination system in Vietnam officially introduced and began administering the Respiratory Syncytial Virus (RSV) vaccine from the British pharmaceutical manufacturer, GSK. This deployment comes as deteriorating air quality in many urban areas elevates the risk of respiratory diseases among the adult population.
The vaccine rollout realizes a strategic cooperation agreement between the vaccination provider and GSK, an agreement that was signed in London just two months prior. The initial vaccine batches have been transported and stored in cold chain systems that meet international Good Storage Practice (GSP) standards.
Vaccine Details and Public Health Impact
This vaccine product was specifically developed for the demographic aged 60 and older and individuals suffering from chronic illnesses. The vaccine functions by utilizing a specific RSV virus protein, combined with an enhancing adjuvant. This mechanism is designed to generate an optimal immune response, helping older individuals—even those with frail health or underlying conditions—recognize and fight the virus effectively and sustainably.
Clinical data indicates the vaccine achieves an efficacy rate of nearly 95% in older adults with pre-existing health conditions. The solution has been approved in nearly 70 countries and has seen over 10 million doses administered globally. In Vietnam, specialized medical organizations focused on Cardiology and Respiratory Medicine have recommended RSV vaccination as an integral part of the management strategy for prevalent chronic diseases such as Chronic Obstructive Pulmonary Disease (COPD) and heart failure.
Burden of Disease Requiring Control
The RSV virus is identified as a primary agent causing severe respiratory conditions, including pneumonia and respiratory failure across all age groups, posing a particularly high risk to the elderly. In this demographic, RSV can lead to critical complications, exacerbate existing health issues, and increase the risk of dangerous cardiovascular events such as stroke or myocardial infarction, potentially resulting in death.
Statistics show that the disease burden caused by RSV in Vietnam is significant. It is estimated that over a five-year period, the virus may have caused approximately 4.6 million infections, 200,000 hospitalizations, and 18,000 deaths in the over-60 age group.
Medical experts state that there is currently no specific treatment for RSV. Therefore, active prevention through vaccination plays an especially crucial role. The vaccine, administered as a single dose, is considered a simple, effective measure to help reduce the risk of hospitalization, severe complications, and alleviate strain on the healthcare system.